The global 3D printing medical devices market is expected to reach $ 1.9 Billion by 2023 from $ 0.9 Billion in 2018, at a CAGR of 18.0% during the forecast period, 2018-2023.
Medical equipment is expensive and result in an increased healthcare spending. According to the Advanced Medical Technology Association, in 2013, the estimated total spending on medical devices and in-vitro diagnostics in US was $ 171.8 billion i.e. 5.9% of the of total national health expenditures. This is one of the major drivers for the 3D printing devices market as, 3D printing splints, medical models used before surgeries or other necessary items of care help in cutting healthcare cost to a great extent. Considering this, the medical device industry is rapidly adopting 3D printing technology to introduce novel products that are revolutionizing medical device design and manufacturing.
3D printing in medical field has resulted in both clinical and innovative advancements. Additive manufacturing technologies have been applied to medical equipment, prosthetics, dental products, bio printing, and pharmaceuticals. However, in order to be able to be used for clinical applications, these technological advancements require a FDA approval. However, recently the FDA issued guidelines speed up the approval process for companies engaged in the development of these technologies. Additionally, these companies are also likely to be entitled to research and development tax credits to balance the cost associated with testing, process improvement, and production. These are some of the important factors responsible for driving the growth of this market.
However, some of the factors like socio-ethical concerns related to the use of 3D-Printed Products
and the complex process involved in this are likely to restraint the growth of this market. Irrespective of these restraints the market is likely to grow to reach nearly 1.9 billion by 2023.
The market for 3D printing medical devices is segmented on the basis of components (software and services), equipment (3D printers and 3D bioprinters), materials (plastics, metal & metal alloys powder, bioprinting biomaterials, and others), by technology (electron beam melting, laser beam melting, photopolymerization, droplet deposition (DD) or extrusion-based technologies, and three-dimensional printing (3DP) or adhesion bonding or binder jetting). The 3D-printed medical products market is further segmented based on type (surgical guides, surgical instruments, prosthetics & implants, and tissue engineering products).
Some of the key answers the study intends to provide are-
• Why 3D printing equipment segment is expected to grow at a faster rate?
• What are the impacts of the regulatory environment?
• Which companies having an edge?
• What are the latest technology trends?
Some of the major players dominating the market are Stratasys Ltd., Envisiontec GmbH, 3D Systems Corporation, EOS GmbH Electro Optical Systems, Renishaw plc, Materialise NV, Arcam AB, 3T RPD Ltd.
Concept Laser GmbH, Prodways Group, Carbon, Inc., SLM Solutions Group AG, Organovo Holdings, Inc.
Biomedical Modeling Inc. and Anatomics Pty Ltd.
1 Introduction
1.1 Goal & Objective
1.2 Report Coverage
1.3 Supply Side Data Modelling & Methodology
1.4 Demand Side Data Modelling & Methodology
2 Executive Summary
3 Market Outlook
3.1 Introduction
3.2 Current & Future Outlook
3.3 DROC
3.3.1 Drivers
3.3.2 Restraints
3.3.3 Opportunities
3.3.4 Challenges
3.4 Market Entry Matrix
3.5 Market Opportunity Analysis
3.6 Market Regulations
3.7 Pricing Mix
3.8 Key Customers
3.9 Value Chain & Ecosystem
4 Demand Market Analysis
4.1 3D Printing Medical Devices Market, By Components
4.1.1 Software
4.1.1.1 Market Size & Forecasts, 2018-2025
4.1.2 Services
4.1.2.1 Market Size & Forecasts, 2018-2025
5 3D Printing Medical Devices Market, By Equipment
5.1 Introduction
5.2 3D Printers
5.2.1 Market Size & Forecasts, 2018-2025
5.3 3D Bioprinters
5.3.1 Market Size & Forecasts, 2018-2025
6 3D Printing Medical Devices Market, By Materials
6.1 Introduction
6.2 Plastics
6.2.1 Market Size & Forecasts, 2018-2025
6.3 Metal & Metal Alloys Powder
6.3.1 Market Size & Forecasts, 2018-2025
6.4 Bioprinting
6.4.1 Market Size & Forecasts, 2018-2025
6.5 BioMaterials
6.5.1 Market Size & Forecasts, 2018-2025
7 3D Printing Medical Devices Market, By Technology
7.1 Electron Beam Melting Materials (Plastics, Metal & Metal Alloys Powder, Bioprinting Biomaterials), Technology (Electron Beam Melting, Laser Beam Melting, Photopolymerization, Droplet Deposition (DD) - Global Market Trends Upto 2025
7.1.1 Market Size & Forecasts, 2018-2025
7.2 Laser Beam Melting
7.2.1 Market Size & Forecasts, 2018-2025
7.3 Photopolymerization,
7.3.1 Market Size & Forecasts, 2018-2025
7.4 Droplet Deposition (DD)
7.4.1 Market Size & Forecasts, 2018-2025
8 3D Printing Medical Devices Market Analysis, By Region
8.1 North America
8.1.1 U.S.
8.1.2 Canada
8.1.3 Mexico
8.2 Europe
8.2.1 Germany
8.2.2 Italy
8.2.3 France
8.2.4 UK
8.2.5 Rest of Europe
8.3 Asia Pacific
8.3.1 China
8.3.2 Japan
8.3.3 India
8.3.4 Australia
8.3.5 Rest of APAC
8.4 Middle East & Africa
8.4.1 Saudi Arabia
8.4.2 UAE
8.4.3 Rest Of MEA
8.5 South America
8.5.1 Brazil
8.5.2 Argentina
8.5.3 Rest of South America
9 Supply Market Analysis
9.1 Strategic Benchmarking
9.2 Market Share Analysis
9.3 Key Players
9.3.1 Philips
9.3.2 Garmin
9.3.3 Omron
9.3.4 Drägerwerk
9.3.5 Nokia Technologies
9.3.6 Jawbone
9.3.7 Polar Electro
9.3.8 World Global Network
9.3.9 Activeinsights
9.3.10 Vitalconnect
9.3.11 Xiaomi
9.3.12 Misfit
9.3.13 Monica Healthcare and Others
SDMR employs a three way data triangulation approach to arrive at market estimates. We use primary research, secondary research and data triangulation by top down and bottom up approach
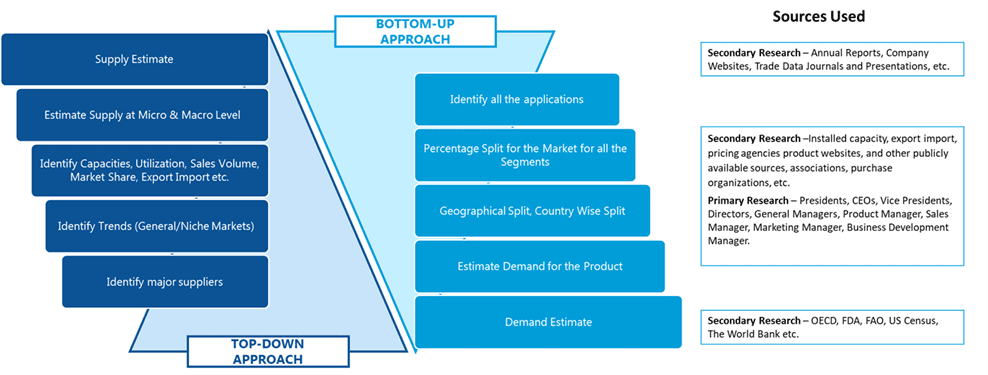
Secondary Research:
Our research methodology involves in-depth desk research using various secondary sources. Data is gathered from association/government publications/databases, company websites, press releases, annual reports/presentations/sec filings, technical papers, journals, research papers, magazines, conferences, tradeshows, and blogs.
Key Data Points through secondary research-
Macro-economic data points
Import Export data
Identification of major market trends across various applications
Primary understanding of the industry for both the regions
Competitors analysis for the production capacities, key production sites, competitive landscape
Key customers
Production Capacity
Pricing Scenario
Cost Margin Analysis
Key Data Points through primary research-
Major factors driving the market and its end application markets
Comparative analysis and customer analysis
Regional presence
Collaborations or tie-ups
Annual Production, and sales
Profit Margins
Average Selling Price
Data Triangulation:
Data triangulation is done using top down and bottom approaches. However, to develop accurate market sizing estimations, both the methodologies are used to accurately arrive at the market size. Insert Image

